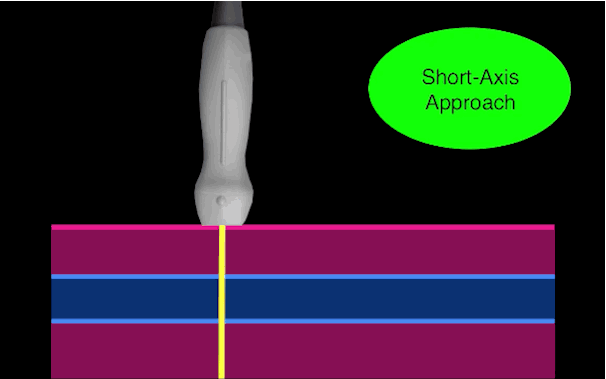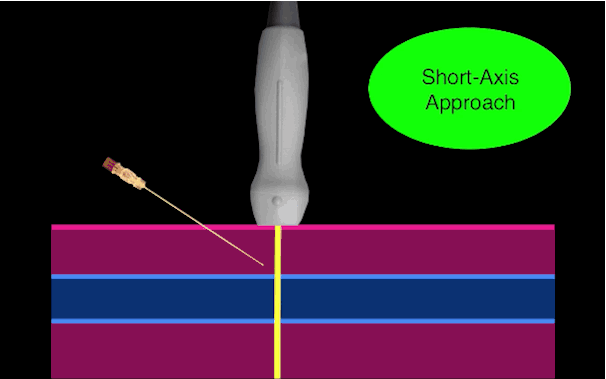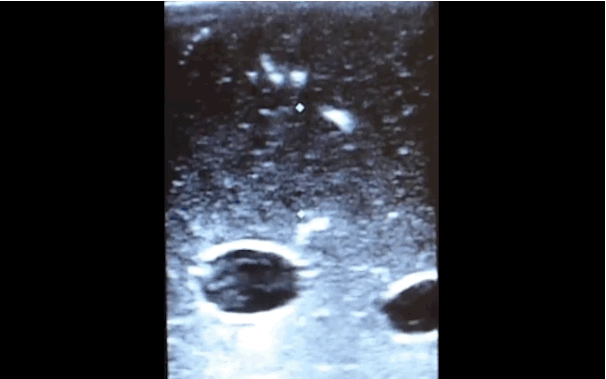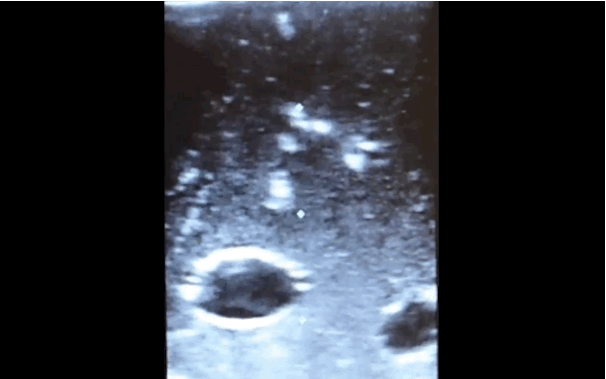
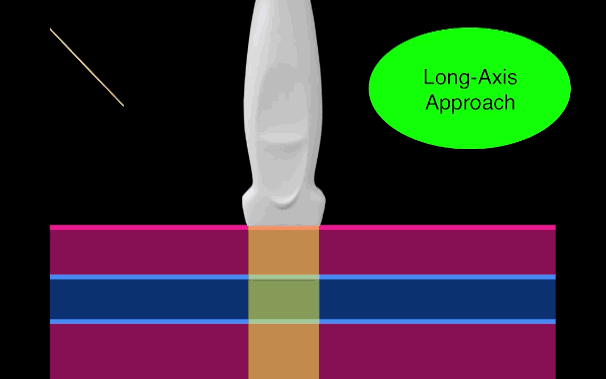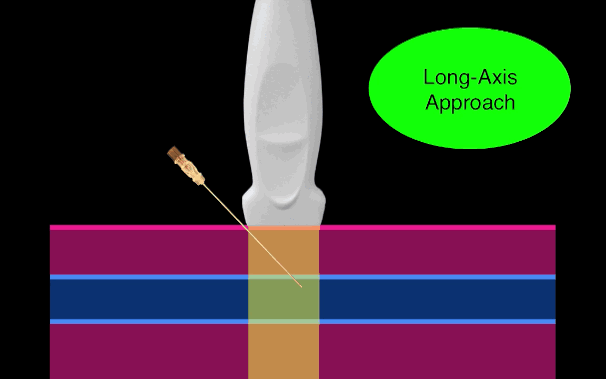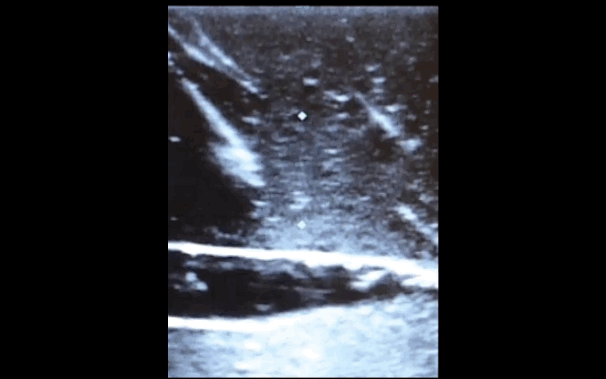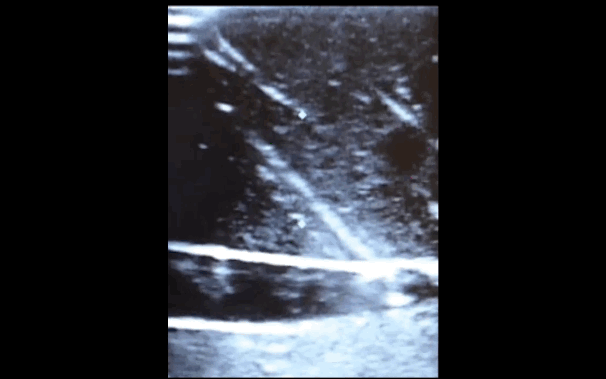
There is little debate that ultrasound-guided central lines are safer, faster, and more reliable compared to a landmark technique; there is some debate, however, as to whether the short axis (SA) or long axis (LA) approach is the best (see clips below).
The referenced study compared the SA and the LA technique for both the internal jugular (IJ) and subclavian (SC) venous approach. The authors measured number of skin breaks, number of needle redirections, and time to cannulation for each method.
This study demonstrated that the LA technique for subclavian placement had fewer redirections, decreased cannulation time, and fewer posterior wall punctures as compared to the SA. With respect to the IJ approach, the LA was also associated with fewer redirections than the SA view.
Bottom line: Consider the long-axis technique the next time you place an ultrasound guided central line.